What are protein domains?
Protein domains are the structural units that comprise a protein. They are often conserved structures that may function independently of the rest of the protein. Domains often serve to be crucial for a specific function or interaction of a protein. Many proteins are made up of multiple domains. These domains are found in various biological contexts and can be arranged differently in a protein to function differently [1].
Protein domain database sites are often used to help identify conserved domains, functional sites, and patterns associated with a protein of interest. The identification of certain domains and the families that they belong to, may provide insight into the function of the protein of interest. These sites include, but aren't limited to, SMART, Prosite, and Pfam.
Protein domain database sites are often used to help identify conserved domains, functional sites, and patterns associated with a protein of interest. The identification of certain domains and the families that they belong to, may provide insight into the function of the protein of interest. These sites include, but aren't limited to, SMART, Prosite, and Pfam.
What domains are found in the AAGK2 protein?
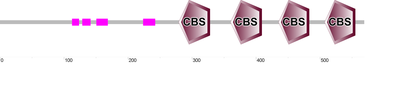
Both Prosite and SMART databases identified four significant cystathionine beta-synthase (CBS) domains in the AMP-activated protein kinase gamma2 subunit. These CBS domains are evolutionarily conserved, small intracellular modules that are present in all three domains of life [2,3]. CBS domains usually come in pairs and are often found in cytosolic and membrane proteins [3].
What are the functions of CBS domains?
|
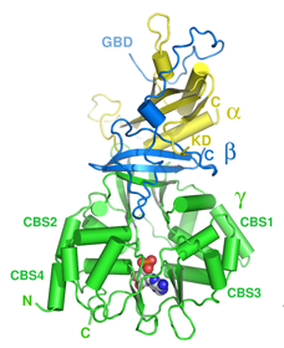
CBS domains often have very different functions, such as acting as metabolic enzymes and transcriptional regulators to being involved in ion channels and transporters, due to the range of proteins they are found in. Although these domains may function very differently, each has the conserved β1-α1-β2-β3-α2 protein folding pattern.
CBS domains that come in tandem pairs (such as the ones found in the AAKG2 protein) are believed to act as binding sites for adenosine derivatives that help regulate the activity of the domain. It has been proposed that the CBS domains found in the AMP-activated protein kinase specifically functions in restoring cellular ATP balance during periods of metabolic stress. Mutation to one of these CBS domains, in humans, often causes a variety of hereditary diseases. Wolff-Parkinson-White syndrome is one of these hereditary diseases, and it's phenotype is seen when there is a mutation specifically in one of the CBS domains of the AMPK gamma2 subunit [3]. |
References
[1] http://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
[2] Bateman, A. (1997) The structure of a domain common to archaebacteria and the homocystinuria disease protein. Trends Biochem Sci. 22(1):12-3.
[3] Ignoul, S., Eggermont, J. (2005) CBS domains: structure, function, and pathology in human proteins. American Journal of Cell Physiology. 289(6):1369-78. doi: 10.1152/ajpcell.00282.2005
[2] Bateman, A. (1997) The structure of a domain common to archaebacteria and the homocystinuria disease protein. Trends Biochem Sci. 22(1):12-3.
[3] Ignoul, S., Eggermont, J. (2005) CBS domains: structure, function, and pathology in human proteins. American Journal of Cell Physiology. 289(6):1369-78. doi: 10.1152/ajpcell.00282.2005
Margaret Beatka ([email protected])
Page Last Updated: 5/10/13
This web page was produced as an assignment for Genetics 677, as an undergraduate course at UW-Madison.
Page Last Updated: 5/10/13
This web page was produced as an assignment for Genetics 677, as an undergraduate course at UW-Madison.